Dashing through the snow, on an Omicron sleigh...
The CDC allows healthcare workers back to work after 7 day quarantines to help combat staffing shortages. Meanwhile, Philips claims that volatile organic compounds from its DreamStation aren't harmful
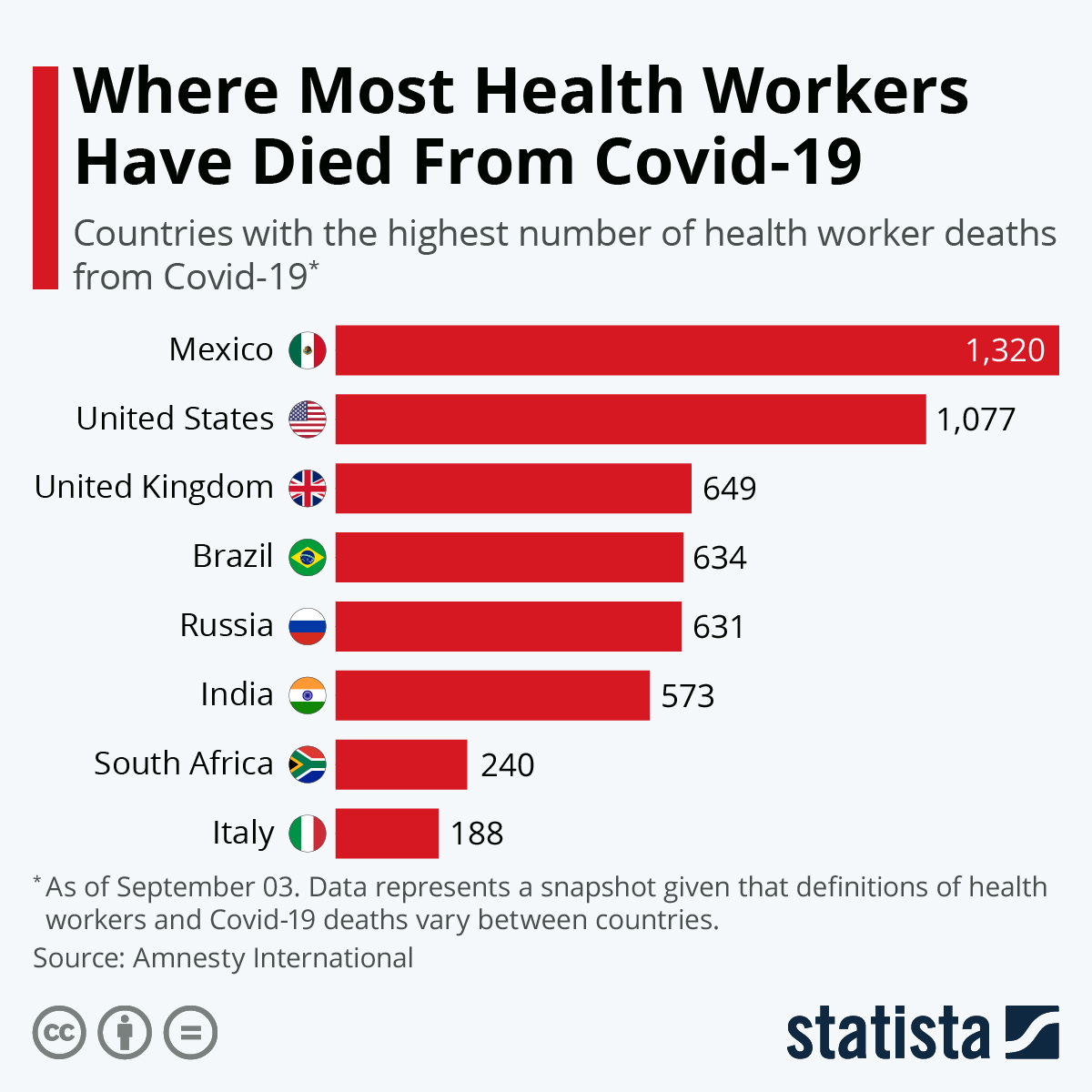
We’re number 2… for killing healthcare workers with covid
CDC Changes Isolation Guidelines for Healthcare Workers
Amid a new wave of coronavirus cases, the Centers for Disease Control and Prevention (CDC) is altering quarantine rules for healthcare workers. Workers that test positive for COVID-19 can now return to work after seven days of isolation instead of 10.
This new rule is meant to help combat the stress the new Omicron cases will bring to already understaffed hospitals.
The specific rule says that healthcare workers may return to work after seven days if they are asymptomatic and test negative. In a statement Thursday the CDC said, "isolation time can be cut further if there are staffing shortages."
The new guidelines also highlight that a negative test result should come within 48 hours of going back to work and could also apply to “mildly symptomatic” staff. Healthcare workers will also not be required to quarantine after high risk exposures if they have all the vaccines, including the booster.
These new guidelines will only apply to healthcare workers and not the general public. There are no plans, as of now, to shorten the isolation period for non-healthcare workers that test positive.
Biogen Cuts Price, Begs People to Take Aduhelm
On Monday, Biogen halved the price of its new Alzheimer’s drug Aduhelm to $28,200. The first new treatment for Alzheimer’s in nearly 20 years, Aduhelm has underwhelmed in sales. In July and September, Biogen sold only a paltry $300,000 worth of Aduhelm. These sales numbers fell far short of the forecasts of more than $12 million and Biogen’s expectation that Aduhelm would become a commonly used drug.
There are two reasons why doctors have been hesitant to prescribe Aduhelm to patients. The main barrier has been Aduhelm’s $56,000 annual per patient price tag. But at the same time, there have been questions about Aduhelm’s efficacy in treating Alzheimer’s. The FDA’s accelerated approval of the drug went against the decision of 10 of the 11 members of its advisory panel, a decision that prompted the resignation of 3 experts on the panel. The FDA, which initially approved the drug for all Alzheimer's patients, now recommends its use only in patients with mild cognitive impairment or mild dementia. In September, one patient died after Aduhelm treatment, prompting further caution from physicians.
Aduhelm’s price cut comes before a January 2022 decision by the US Medicare program on whether the use of the drug will be covered by Medicare. The earlier $56,000 price tag had been viewed as a serious burden on Medicare and the US tax payer, with projected annual costs of between $6 billion and $29 billion. Aduhelm’s new price will change the decision making process for physicians, patients, and regulators.
Philips Argues Recalled Devices Are not Harmful
Over this year, the medical technology giant Philips has been faced with several recall orders. One of the largest ones being for its sleep apnea device called the DreamStation.
Sleep apnea is a disorder where a sleeping person’s breathing repeatedly starts and stops. This can be fatal so devices like the DreamStation are usually recommended to help the patient breathe.
However, the DreamStation was shown to increase a patient's exposure to volatile organic compounds (VOCs). This was the argument for their recall. But in an update this past Thursday, Philips has stated that DreamStation related VOCs are not typically associated with long term health consequences.
This means they are admitting that the DreamStation exposes the patient to VOCs. Furthermore, the company is also declining to comment on any short or medium term consequences of using the DreamStation. It is possible that Philips’s claim about there being no long term consequences is true, however “long term health consequences” encompasses many things and, depending on interpretation, may not include minor but persistent side effects like a constantly dry throat or some other non-lethal, nor particularly disruptive symptom.
Featured Fake News
This image has been circulating on Facebook and Twitter this past week:
Well, it’s completely fake. No such thing was published anywhere in the New York Times. We haven’t been able to find it, Reuters couldn’t find it, and the New York Times denies ever having such a thing on their site and website archives seem to agree.
So no, teachers are not allowing vaccinated kids to bully unvaccinated kids. Although if something like this is circulating, teachers can and should expect Karen’s to question them on it.
Thank you for reading! If you are not already subscribed, we would appreciate if you would subscribe using the button below.